The Prevention of Recurrent Bilateral Capsular Contracture with BellaDerm® Acellular Dermal Matrix
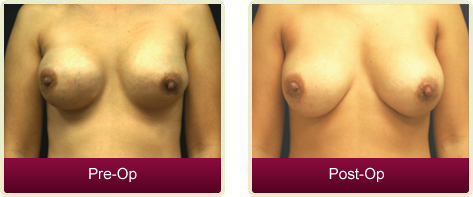
One of the most common complications of breast implant surgery is capsular contracture, with reported rates varying widely between 12 and 30 percent of patients who receive implants. This condition is defi ned as a tightening of the fi brous capsule surrounding a breast implant, resulting in fi rmness or hardening of the breast and causing great pain and discomfort for the patient. The pathology of this complication is not fully understood, but may be the result of subclinical infection or chronic inflammation.
A therapy that has gained attention in recent years is the use of an acellular dermal matrix (ADM) as an interface in the space between the breast and the implant to prevent the formation of a tight, fi brous capsule. It is believed that the proteins remaining in the extracellular matrix of the ADM assist the wound healing process and enable host incorporation and integration. In contrast, implant contact with the native submuscular breast capsule may cause the wound to persist in the infl ammatory phase. Human histopathology studies show a direct correlation between prolonged infl ammation and a thicker capsule formation.
CASE HISTORY
A 40 year old hairdresser presented for evaluation of severe bilateral capsular contracture post breast augmentation. She has a signifi cant history of recurrent capsular contracture, post multiple capsulectomies and replacement of saline and silicone implants over an eight-year period. The last surgery was six months prior with bilateral capsulectomies and new silicone implants (Mentor® 325cc round and smooth high profile) placed submuscularly, but capsular contracture recurred after three months. She had considered just removing the breast implants, but decided, instead, to undergo an additional revision.Procedure
The patient underwent a bilateral capsulectomy and implant with BellaDerm dermal matrix graft. Given the patient's extensive history of recurrent capsular contracture, a minimally processed, human ADM, BellaDerm, was chosen over a xenograft or synthetic graft, to minimize the risk of recurrence due to inflammation or foreign body response. BellaDerm is processed without the use of ETO, gamma irradiation or other means of terminal sterilization (FDA regulations require that all xenografts must be terminally sterilized) so that it remains more like the patient's own tissue. Literature shows that harsh chemical processing may negatively impact cellular bioactivity and delay tissue incorporation, which may, in turn, increase the opportunity for fibrotic capsular formation. Synthetic meshes, while relatively new and unproven in the breast, have demonstrated significant rates of infection in the abdominal wall and pelvis.Once the capsulectomy was performed, an 8cm x 16cm sheet of BellaDerm acellular matrix was then sewn in the inferior half of the pocket on each side. The patient chose to keep her 325cc Mentor silicone implants since they were only six months old. The inferior edge of the graft was sewn to the inframammary fold with 3-0 Vicryl interrupted sutures. The superior portion of the ADM was sutured to the inferior edge of the muscle, also with 3-0 Vicyrl.
A JP drain was placed in each pocket and the skin was closed with 4-0 Monocryl.
Follow-Up
After the drains were removed at one week follow up, both the surgeon and patient agreed the breasts looked and felt much softer. At six months post surgery, the patient continues to have very good results with soft breasts and no visible or palpable evidence of capsular contracture. Continued follow up is planned.Conclusion
The use of acellular dermal matrices has been well described in breast reconstruction, with good results for defining the shape and contour of the reconstructed breast. Although several studies have been published supporting its role in the remediation of capsular contraction, ADM is still less tried in this application. To date, treatment of capsular contractures has not been very successful due to high recurrence rates, particularly in patients with bilateral capsular contracture. In this patient with an extensive history of bilateral capsular contracture, BellaDerm, a non-terminally sterilized ADM, was selected to minimize the risk of recurrence secondary to inflammatory response or infection, and thus far, has proven to be effective. Longer follow up and additional evaluation is needed to clearly delineate the protocol for treatment of capsular contracture.Related Articles
- 7 Things to Know Before Getting Breast Augmentation in Huntington Beach, California
- Los Angeles plastic surgeon explains what you need to know about breast augmentation
- What Are The Pros and Cons of Breast Augmentation Surgery in Los Angeles
- What You Need to Know About Perfect Breast Augmentation in Huntington Beach, CA
- Los Angeles plastic surgeon explains what you need to know about breast augmentation
- Fall in love with your curves with breast augmentation services in Huntington Beach, CA
- Safe breast augmentation from a skilled specialist you can trust in Huntington Beach, CA
- Huntington Beach patients find breast implants are worth it, thanks to cosmetic surgeon’s skill
- Orange County plastic surgeon discusses the various types of breast augmentation surgery
- The Prevention of Recurrent Bilateral Capsular Contracture with BellaDerm® Acellular Dermal Matrix
- Who is the best breast implant surgeon in the Orange County area?
- Why might Orange County area women consider breast implants?
- Techniques of breast augmentation provided by Orange County, CA area plastic surgeon
Back to Breast Augmentation Home Page
 A Magna cum Laude graduate from Millsaps College, Dr. Thomas T. Nguyen is a board certified plastic surgeon. He brings with him a plethora of skill, expertise and knowledge given his 20 plus years of experience in the field.
A Magna cum Laude graduate from Millsaps College, Dr. Thomas T. Nguyen is a board certified plastic surgeon. He brings with him a plethora of skill, expertise and knowledge given his 20 plus years of experience in the field.Dr. Nguyen is presently the medical director of Orange County Cosmetic & Reconstructive Surgery Center. He has engaged in research and contributed to many medical publications and continues to be deeply passionate about providing the best possible treatment for his patients.




